Bond polarity6/24/2023  
1 A dipole is when a bond has a positive and negative charge at either end. This allows metals to give some of their electrons to nonmetals, resulting in a dipole. That means that they have weaker electronegativity than many nonmetals. 
To help determine whether a bond is polar or nonpolar, you must know the. (Credit: Jodi So Source: CK-12 Foundation License: CC BY-NC 3. Metals are typically shiny and malleable. Using the Electronegativities we can predict whether a given bond will be non-polar, polar covalent, or ionic. But if an electron is more attracted to one atom than another, the bond is polar. Electronegativity values tend to increase as you go to the right and up on the table shown in the image below.\): A nonpolar covalent bond is one in which the distribution of electron density between the two atoms is equal. Thus, each B-F bond in BF3 (Boron Trifluoride) molecule is polar. The difference between the electronegativity values of B and F is equal to 1.94 (3.98 2.04 1.94) which is greater than 0.5. To help determine the electronegativity of an atom, look at the periodic table of elements. To determine if a bond is polar or not we must find out the difference between the electronegativity values of the two atoms sharing a bond. Electronegativity is the power of an atom in a molecule to attract electrons to itself. 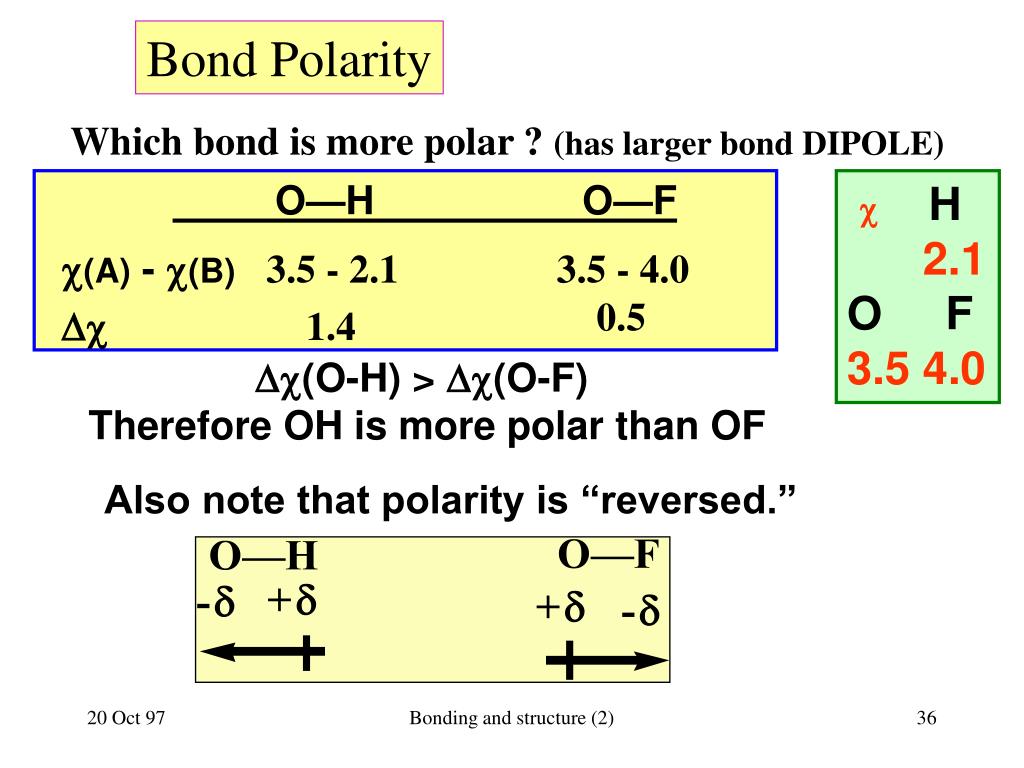
The polarity of a bond depends on the electronegativities of the bonded atoms. Electronegativity is defined as the ability of an atom to attract electrons when the atoms are in a compound. To help determine whether a bond is polar or nonpolar, you must know the electronegativity. Bonds between carbon and other elements such as oxygen and nitrogen are polar. A bond is said to be a polar bond,if it has. Thus, an H-S bond is non-polar, but the molecule overall is polar due to the asymmetric bent shape of the H2S molecule. This is because there is only a slight electronegativity difference between the hydrogen (H) and sulfur (S) atoms bonded in H2S. But if an electron is more attracted to one atom than another, the bond is polar. Polarity in organic chemistry refers to a separation of charge and can describe a bond or an entire molecule. Hydrogen sulfide (H2S) is a weakly polar molecule. If the electron is shared equally between the atoms in a covalent bond, then the bond is nonpolar. Covalent bonds are formed between one or more pairs of shared electrons. Ionic bonds are formed between two oppositely charged ions, such as a metal and a nonmetal. There are two basic types of bonds, covalent and ionic. To know the structure, you must first know how the atoms and molecules are arranged. Specifically, it is found that, while bonds between identical atoms (as in H 2) are electrically uniform in the sense that both hydrogen atoms are electrically neutral, bonds between atoms of different elements are electrically inequivalent. The more electronegative element pulls on the bonding electrons more. The polarity of a bond is the distribution of electrical charge over the atoms joined by the bond. 
Why are some gases greenhouse gases and others are not? The structure of a molecule influences the way it interacts with infrared radiation. Bonds are polar if two elements of differing electronegativities are bonded to each other. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
